SN2 & SN1, E2 & E1. A Simpler Method (Part II)
The Zen of SN2 & SN1, E2 & E1. (Part II)
How to Crush Substitution and Elimination With Total Confidence
If determining what you should do has you guessing or stumped you completely—you may laugh…or possibly cry…when you realize how simple all of this is:
You are normally given an alkyl halide (R-X) which is an electrophile possessing a suitably good leaving group as the substrate. You are doing reactions under condition necessary to get the leaving groups to do what they are there for in the first place. What is that purpose, you ask?
Think about the classes of reaction you’ve covered. It may seem like there are dozens, but there are only about five this semester. You’ll quickly realize that the first semester covers: addition of electrophiles across π-bonds, free-radical chain reactions, select oxidation-reduction reactions, and now substitution and elimination reactions. Halides are good leaving groups and are meant to leave. If you understand this much, you are on your way to understanding the goal. The halides are weak bases, forming weak bonds, and placed on sp3 carbons in your questions. (They are sometimes present in some biological molecules but are not normally meant to leave; and you have no control over that).
Your goal, therefore, is to transform them either a concerted bimolecular substitution/elimination or a multi-step unimolecular substitution/elimination. If you’ve approached the correct solution, the halogen atom will be gone when you are finished.
Why is This Confusing for Most?
At all times, even in all textbooks, two reaction chapters are presented and treated as though they are competing. Two substitutions, SN2 and SN1 are contrasted, and is followed by a chapter on eliminations, E2 and E1.
These reactions, though they invariably are, should not be presented this way. You are much more likely to become confused when the choices seem to be between SN2 vs SN1; and likewise when to decide between E2 vs. E1, in the following chapter.
It seems the two limiting (limiting meaning extreme, or characteristically ideal) substitution reactions (SN2 vs. SN1) appear to be competing! Therefore, the two limiting elimination (E2 vs. E1) reactions are competing as well. This is a confusing headache.
The reality is that the reaction you will do—if one is possible—will be either a bimolecular or a unimolecular reaction but never both.
Is this becoming clear?
The limiting substitution reactions at sp3 carbon are covered in textbooks together this way as though you are learning how to contrast the subtleties between each substitution and then differentiating between them all or, just as mistakenly, identifying one perfect reaction. This is NOT the case. People start assuming you are learning to decide between all of them. It is assumed there is always one answer—or four of them! This is never the case, as you don’t have a choice.
Confused? It confuses the majority of students.
Think about the following if you want to propose a correct solution:
-
You will never do both Substitution reactions (SN2 and SN1) on the same compound under the same conditions.
-
Likewise, you will never do both Elimination reactions (E2 and E1) on the same compound under the same conditions.
Why the SN1 and the SN2 are ever been presented in the same chapter makes no sense. Some will say, well, they’re both substitutions and should be grouped together. Hogwash! Regardless of the outcome, the SN1 and the E1 have far more in common then the SN2 and the E2. This long-standing tradition promotes general or total confusion. Don’t mix kinetically distinct mechanisms.
To jumble unimolecular and bimolecular reactions with each other makes no sense in the mind of the the learner or to anyone else . The conditions of the reaction (the reagents above and below the arrow) make only one type of reaction kinetically feasible. The route to solving the problem has always been present.
The outcome is dependent on kinetics and rate laws.
Despite the confusing tables of unnecessary information out there (which you paid to confuse yourself with) you are not deciding between all 4 reactions, just whether it is bimolecular or unimolecular. Then draw all the substitution and elimination products for the likely kinetics. If both substitution and elimination are possible, for whichever pathway is indicated, do them both since both are correct answers.
Whether it’s one-step or more, attempt both unimolecular reactions SN1 and/or E1 OR attempt SN2 and E2: never both! If they are possible determine which elimination reaction is major using Zaitzev’s rule. If it is not a tertiary (or neopentyl) halide, attack the backside. Then fill in the blank. That’s it, you are done.
The majority of people are confused about this believing it is more complicated than it is. I have seen people allow this to make this into a bigger deal than it is. The cognoscenti will gripe about this or that…”the prototypical SN2 is defined using methanol…etc.” True but that is one experiment which, by the way, is beyond the scope of undergraduate organic chemistry.
It is not a competition between SN2 vs SN1 and then deciding between E2 vs. E1. It is a competition between SN2 & E2 or SN1 & E1. In practice this has a simple outcome.
The question you should ask yourself every time you face such questions is: Is the reaction unimolecular or bimolecular?
Once you correctly make this determination, draw the mechanisms of all the possible reactions, then determine which products are major. Is it E2 / SN2 (concerted = one step or “synchronous”)? OR is the reaction E1 / SN1 (multi-step involving a carbocation intermediate)?
That is the question to ask. Once you know the answer to that question, attempt to do both on the R-X substrate.
How Can I Know the Difference Between Unimolecular and Bimolecular?
The “2” in bimolecular and the “1” in unimolecular have kinetic significance. You will almost never be given that information. You’ll be given something else instead…a reagent.
The rate of the bimolecular reaction is concentration-dependent and is the faster reaction as it is the one that has kinetically unstable molecules colliding and reacting because they want something. They want to become stable but are missing something (hence the terms containing “-phile“).
The rates of these reactions is optimized in the lab so the concentrations are high but the learner doesn’t see this doing “chemistry on paper”. An anion (negatively charged nucleophile or base) wants a cation, a proton or an electrophilic (+ charged) center and vice versa.
On the other hand, the unimolecular reaction with a neutral reactant (it’s the solvent), therefore cannot and does not compete in rate with a bimolecular reaction which will leave it in the dust. The unimolecular processes are not an option to arrive at the major product when stronger species are present. Textbooks say a bimolecular reaction is favored by a “high” concentration of good nucleophile or strong base. Again, this is the type of ambiguity that creates confusion.
How much is a “high enough”concentration? It would be wise to use at least an “equivalent” or “equimolar” amount, wouldn’t it? Mole to mole; one to one or even an slight excess of nucleophile: That would be a high enough concentration. You would not do less if you wanted one result (it wouldn’t be there at all if it were not high enough in concentration). No chemist in their right mind would ever add an less than high enough concentration of something. Are you beginning to see how, well, stupidly things are phrased in your textbook?
How Do you Determine Whether the Reaction is Bimolecular or Unimolecular?
Don’t assume you have a choice among all of the reactions. You don’t. If you assume you do, you will get confused about strength of base, the ‘goodness’ of the nucleophile, and whether a solvent is protic or not, and so on to rule out other irrelevant details. In this class, if the reagent is negatively charged, the reaction will be bimolecular. It’s in salt form: NaOH, NaOMe, NaSH, KOH, KOt-Bu, NaCN, NaN3, NaNH2….etc. you can just ignore (cross out the metal cation actually) the sodium (Na+), lithium (Li+) or potassium cation (K+).
These Group I alkali metal cations (but not hydrogen, which is the oddball of the periodic table and is unlike the elements in its group) are just a placeholder or spectator. Many question-writers omit the cation assuming this is doing you some kind of favor. They are awful awful people.
Think about it: These people are actually setting you up to fail any standardized exam that involves organic chemistry and these common reactions. Standardized exams do NOT leave out cations. It’s you’re job to learn to ignore them. Even worse, I know many professors who just tell you what reaction to do. You will never learn anything that way!
The anions are reactive having negative charge excess. You are focusing on what the negatively charged species does in a bimolecular reaction (Nu or Base). The starting material is an electrophile with a good leaving group, R-X (X=Br–, Cl–, I–, –OTs, –OMs, –OTf).
Under the reaction conditions, whatever is provided in the question is enough to say which substitution or elimination mechanism it will follow and whether it will occur in just one step, or more than that. If you just are given water, that’s your nucleophile and/or base. Being non-charged, it is a poor/weak one making the reaction unimolecular. Is water strong? It’s neutral (weak), so you must do SN1 or E1, and if you can’t do either of those (you will rarely, if ever, be given such questions unless it’s a trick question (yes, there are instructors who believe organic chemistry isn’t already difficult enough). The answer is: nothing changes, “no reaction”, or “N.R.”.
The Substrate Tells you the Rest
Is a methyl cation going to undergo solvolysis to undergo a unimolecular reaction? Of course not. First it has to become a carbocation, and you can’t make unstable methyl cations without a much stronger catalyst to cause it to form. This one is common sense: you cannot make an alkene from (eliminate) a methyl halide because…? You need two carbons to have or create a double bond. The starting material sometimes tells you what reaction you can or can’t do.
But the reagent, without exception, tells you can do both of either bimolecular reaction if the reagent is strong (reactive and negatively charged). You will do a unimolecular if it is weak (neutral and slow to react). “Okay, what if it’s an alcohol in strong acid?” There are no negatively charged species in acidic solution. So what do you think?
Even simpler put:
If the reagent is just an alcohol (R-OH) or water (H2O) it’s going to be SN1 / E1. TRY IT. You’ll see it is always correct even though this plain fact isn’t talked about anywhere where you will find it. Even people who think it’s more difficult than this, will concede you are correct: do enough problems, look through enough answer keys, and you will see it.
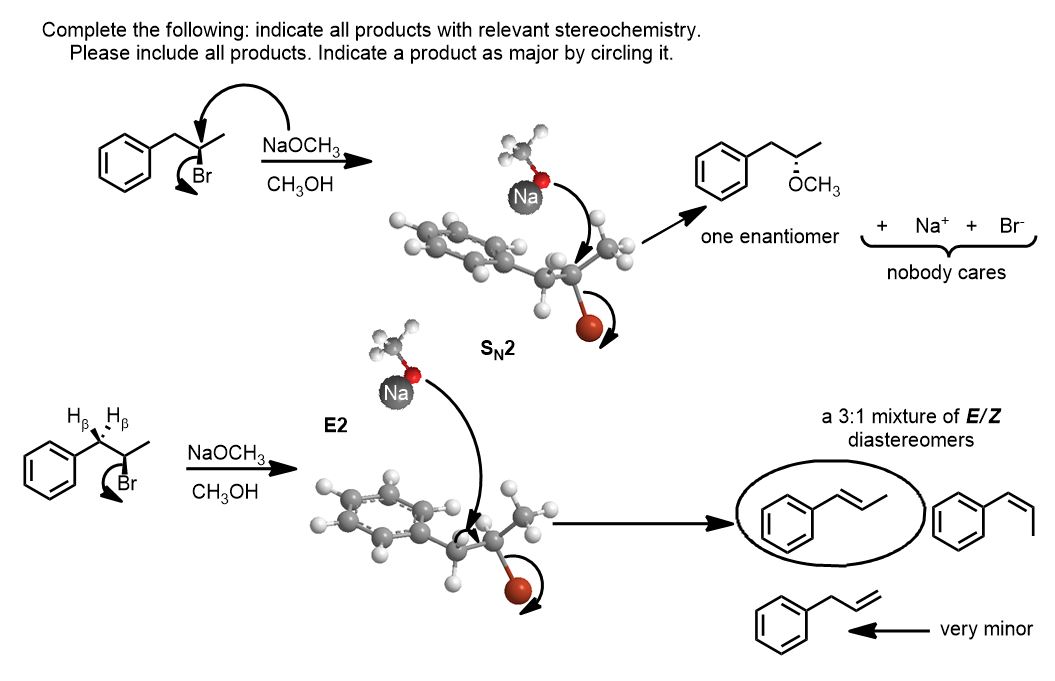
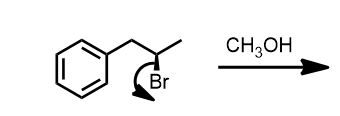
Let’s say you are given the following problem. How would you solve it?
Based on the Reagents alone: Is the reaction SN2/E2 or SN1/E1?
Is the nucleophile good and/or is the base strong? Sodium methoxide anion is both an excellent nucleophile (substitution) and a strong base (elimination). How do you know that it is good/strong? This reaction must be bimolecular. It must be E2 and/or SN2 because the reagent is strong—meaning not neutral…it’s quite polar.
That is all you can and must do. All you know is whether you have a good nucleophile or strong base. (Yes there’s a protic solvent that does not make it unimolecular (non-bimolecular). Solvent is not so important if a negatively charged species is present. A high concentration of good nucleophile and or strong base (negatively charged) is what determines the reaction’s kinetics. There is a gray area for people when the alkyl halide is secondary. That should never be a concern. You should and can know before starting to draw the mechanism, whether it is unimolecular or bimolecular.
(Substitution) RateSN2= ksub [R-X][Nu-]
(Elimination) RateE2 = kelim[R-X][Base-]
What better solvent than methanol for this reaction? It’s the perfect solvent. But don’t get confused. Does it react faster or slower than sodium methoxide? That’s the primary question.
It is the solvent, the swimming pool it is floating in. Neutral solvent react much slower than negatively charged methoxide and this makes sense. If it is neutral and has no charge it just undergoes diffusion and waits for H-bonding to cause ionization. It’s concentration is not even part of the rate law for the reaction.
(Substitution) RateSN1 = ksub [R-X]
(Elimination) RateE1 = kelim [R-X]
Sodium methoxide is a strong base and an excellent nucleophile being negatively charged and is waiting to collide into something to become stable (neutral). Sodium is not a hydrogen atom which is covalently bonded and can never be ignored. Na+ it is just a spectator that stabilizes the negative charge by coulombic attraction and stabilization.
You cannot compare bimolecular and unimolecular rates like that. There is a huge difference in rate. One is fast and highly energetic (bimolecular and concerted); the other is slow (unimolecular and dependent on carbocation stability). Solvolysis, the first step of both unimolecular mechanisms is rate-determining and it is slow compared to the rate of a bimolecular reaction. How do you know if it’s bimolecular? The reagent is negatively charged.
The exception are amines which are outstanding nucleophiles but uncharged (R3N:)). Does it matter? NO. You won’t react amines in an SN2 (polyalkylation or exhaustive alkylation will occur) and amines aren’t strong enough bases to trigger and E2. Some reactions require exhaustive methylation…but when that’s the case, you will know it already.
If it is Negatively Charged (Anionic in a Salt) it will be a Bimolecular Reaction.
You must do both the SN2 and the E2 reaction if it’s possible.
Now there are further questions.
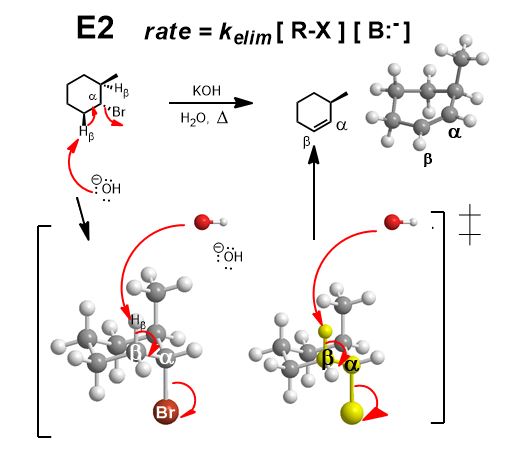
Can you do the concerted substitution? Yes. As long as it’s not a tertiary alkyl halide (or neopentyl) you can attack the back-side. If it’s a chiral center (it is)–draw inverted stereochemistry.
Can you do the elimination? In this example, the Zaitzev product is highly favored because it increases the amount of electron delocalization by resonance.
*That does not mean E2 is always included. Can you do the Elimination? If and only if the base is strong enough to do an E2.
The question is: are -CN, -SH, -SR, -N3, and others basic enough to eliminate H-X? No they aren’t. You won’t do the E2 reaction. While these are excellent nucleophiles, they are insufficient as bases. You’ll substitute from the back side and if you can’t (it’s tertiary or neopentyl and too sterically encumbered) you simply write there is “no reaction.”
Whatever you have that’s negatively charged, it must be as strong or stronger than hydroxide. The conjugate base of water. It is the most familiar strong base you know since high school or general chemistry. So which reactions will do both bimolecular substitution and elimination? NaOH (hydroxide), NaOR (alkoxide) and stronger bases.
If the conjugate base is as strong as hydroxide or an alkoxide, it will do elimination. In fact, if the base is any stronger than an oxyanion, (like NaNH2) the reaction will favor elimination alone. If the base is such a strong base (aka “hard“) all it wants is a proton (eliminating H-X, rather than substituting).
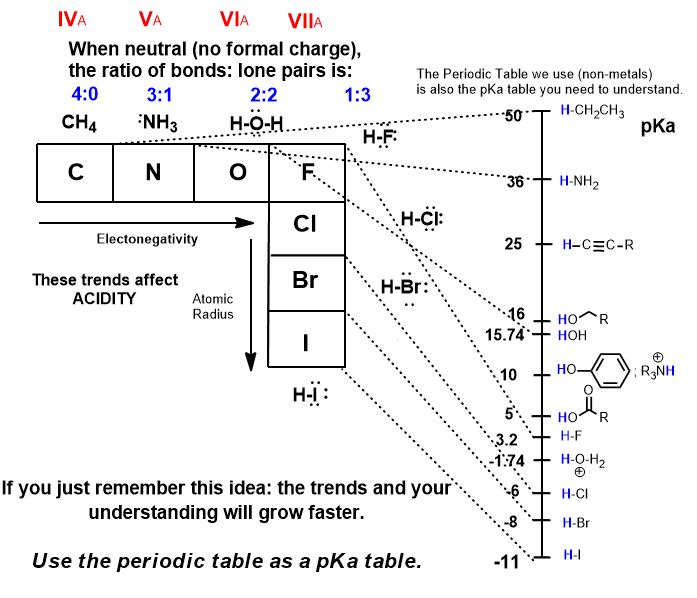
Recall the pKa table. Oh it’s too much to remember?
It is the Periodic Table.
The pKa table you need to know is and always has been no more than the periodic table. SURPRISE!
See Acidity and Basicity.
The lower the pKa (base ten logarithm means each unit is an order of magnitude (10x) difference), the stronger the acid. So what’s the strongest acid in the periodic table we are using? Hydrogen iodide (hydroiodoic acid) has a pKa of -11, and is the strongest acid. That means it’s conjugate base, (iodide anion) the thing it becomes after it loses a proton…it becomes the weakest base there is. After losing it, why would it want it back, to let it go then accept it and make a bond to it? It wouldn’t care to have it back. That’s the idea behind the relationship of acids and their conjugate bases.
The halides (except fluoride) do not form strong bonds to H+ or anything else. That is the definition of acidity. Using the table, what’s the strongest base we will consider? It’s the conjugate base of an alkane. The sp3 C-H bond is so difficult to remove, that we don’t have a base strong enough to remove it. There are indirect methods to make them. Certainly, t-BuLi, n-BuLi, (R-Li); Grignard reagents (R-Mg-X), and other unstabilized carbanions are the strongest bases we will see and use but they are not made in acid-base (proton transfer) reactions.
So that’s it?
Uh, yeah. That’s it. If the reagent is ionic and has a negative charge. Ignore the cation, of course, it is charged but never going form a covalent bond to anything. The positive charge we care about is on carbon if it exists. If not, we hardly pay attention. Na+, K+, +Mg-X, Do this for all metals from now on even when you get to organometallic reactions. You were ignoring them because they are spectator ions and do nothing of consequence, while this is true of the main group metals, the transition metals, on the other hand, can’t be ignored. They are truly incredible in terms of how they catalyze a reaction that doesn’t seem possible.
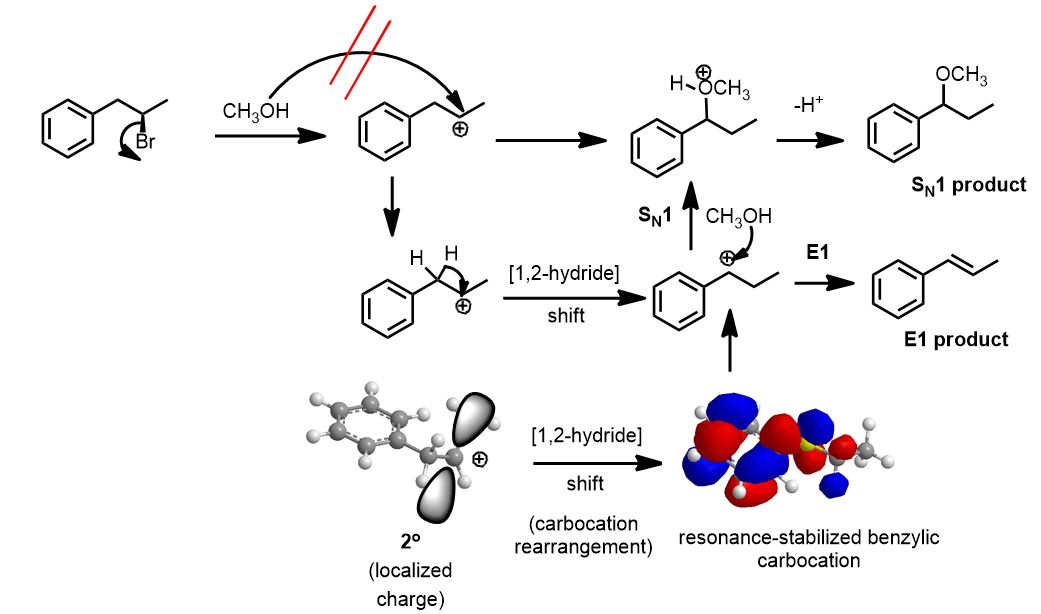
What if it was just methanol (neutral)?
If it’s just methanol then it’s weak. Unimolecular reactions only are possible and start with the rate-determining first step of solvolysis. The leaving group just departs in an ionization process to give you a carbocation.
The reagent is neutral (weak). The bond between oxygen and H is not ionic. It is covalent. Water and alcohols are neutral. You can only do an SN1 or E1 reaction in such a case. Unless told otherwise, you will do both and draw all products for the SN1 and E1 reactions. Circle the major one or put an ‘X’ through the minor ones.
The thing about this carbocation you get (2o), it’s just not as stable as one that might exist next door (2o benzylic). You must do a hydride shift to get the major substitution product. The E1 product is the same whether the carbocation rearranges or not: you therefore get the conjugated alkene (stabilized by benzene).
These are both solutions to both unimolecular substitution and elimination reactions and, unless you are asked for just one, you must include both. Both take place on paper so, as always, write them all out then make a decision, damn it.
A self-respecting chemist will avoid any unimolecular reaction if possible. Not only does it create racemic mixtures of substitution products at any chiral center, the elimination products can potentially come from rearranged intermediates. If it’s thrust upon you however, you have no option but to just do it, as above. People (not necessarily you) make this way too hard for no reason or just have no soul and are terrible people. Seriously.
Rate Laws for the Unimolecular Reactions. When the solvent is water, an alcohol, or a carboxylic (invariably acetic) acid:
Rate = kSN1 [R-X]
Rate = kE1 [R-X]



Leave a Reply
You must be logged in to post a comment.